MCAT Organic Chemistry Question 110: Answer and Explanation
Home > MCAT Test > MCAT organic chemistry practice tests
Test Information
- Use your browser's back button to return to your test results.
- Do more MCAT organic chemistry practice tests.
Question: 110
5. What is the final product of the following reaction?
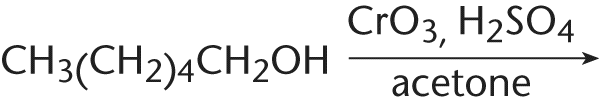
- A. CH3(CH2)4CHO
- B. CH3(CH2)4COOH
- C. CH3(CH2)4CH3
- D. HOOC(CH2)4COOH
Correct Answer: B
Explanation:
Jones reagent (chromium trioxide in aqueous sulfuric acid) is an oxidizing agent. As such, it oxidizes primary alcohols directly to carboxylic acids. This reagent is too strong an oxidant to give an aldehyde, so choice (A) is incorrect; remember that pyridinium chlorochromate (PCC) is a common oxidizing agent used to convert alcohols to aldehydes without progressing to a carboxylic acid. Choice (D), a dicarboxylic acid, cannot form because there is no functional group on the other end of the molecule for the reagent to attack, and it cannot attack an inert alkane. Choice (C) represents reduction, not oxidation.
