MCAT General Chemistry Question 258: Answer and Explanation
Home > MCAT Test > MCAT general chemistry practice tests
Test Information
- Use your browser's back button to return to your test results.
- Do more MCAT general chemistry practice tests.
Question: 258
7. Nitrate is best described by a resonance average of three structures:
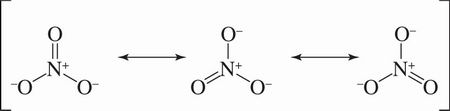
What best describes the peaks in an IR spectrum that result from the three N—O bond stretches?
- A. One peak at the double bond N=O stretch frequency and two peaks at the single bond N—O stretch frequency
- B. One peak at the double bond N=O stretch frequency and one peak at the single bond N—O stretch frequency
- C. One peak at the double bond N=O stretch frequency and one peak between the single N—O and double bond N=O stretch frequencies
- D. One peak between the single N—O and double bond N=O stretch frequencies
Correct Answer: D
Explanation:
D A molecule essentially exists as an average of its resonance structures. No individual resonance structure exists in isolation. In nitrate there are three bonds of equal strength, containing about 33% N=O double bond character and about 67% N—O single bond character. Therefore, there will be only one stretch peak in the IR spectrum intermediate to the N—O and N=O stretches that accounts for all three of these bonds in nitrate.
