MCAT General Chemistry Question 123: Answer and Explanation
Home > MCAT Test > MCAT general chemistry practice tests
Test Information
- Use your browser's back button to return to your test results.
- Do more MCAT general chemistry practice tests.
Question: 123
3. Two organic liquids, pictured in the figure below, are combined to form a solution. Based on their structures, will the solution closely obey Raoult's law?
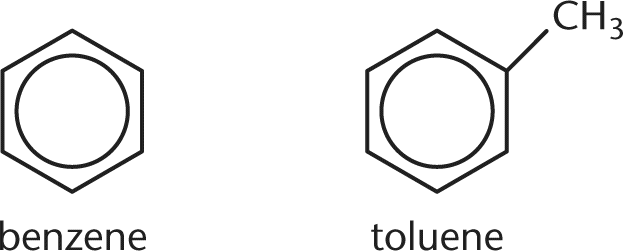
- A. Yes; the liquids differ due to the additional methyl group on toluene and, therefore, will not deviate from Raoult's law.
- B. Yes; the liquids are very similar and, therefore, will not deviate from Raoult's law.
- C. No; the liquids differ due to the additional methyl group on toluene and, therefore, will deviate from Raoult's law.
- D. No; the liquids both contain benzene rings, which will interact with each other and cause deviation from Raoult's law.
Correct Answer: B
Explanation:
Benzene and toluene are both organic liquids and have very similar properties. They are both nonpolar and are almost exactly the same size. Raoult's law states that ideal solution behavior is observed when solute-solute, solvent-solvent, and solute-solvent interactions are all very similar. Therefore, benzene and toluene in solution will be predicted to behave as a nearly ideal solution.
